Do testosterone distributions really overlap between men and women?
A highly cited academic paper made the bizarre claim of a “considerable overlap” in testosterone levels between men and women. I debunk this claim and speculate on how it originated.
📢 Claim
In 2018, five professors of psychology published an academic article arguing that scientific research has “fundamentally undermined” the notion of the sex binary, which posits that humans are a sexually dimorphic species with males and females as the two sexes.1 Five “challenges”, supposedly based on empirical findings from five different disciplines, were presented by the professors to support their argument. As of April 24, 2026, that article has been cited more than 1,500 times, mostly in support of the central thesis that sex is not binary, and many times in support of a specific claim about testosterone.
This claim was that testosterone distributions showed a “considerable overlap” between men and women. It was made in the section of the paper that was probably authored by Sari van Anders of Queen’s University in Canada, because that section contains numerous citations of her work, and also because she made the same claim in other articles.2 Here is the original text:
During adolescence, testosterone levels increase in both boys and girls, but at a much higher average rate for boys. However, the size of this difference has been mischaracterized; although testosterone levels are higher in men than women, on average, the difference is much smaller than widely believed and the distributions show considerable overlap.3
In this article, I briefly examine the truth of this claim (spoiler alert: it’s false), and I dig deeper in an attempt to identify its possible origins.
❌ Refutation
The claim that there is a “considerable overlap” between men and women in the distribution of testosterone levels is astonishing given the considerable existing evidence that refutes it.
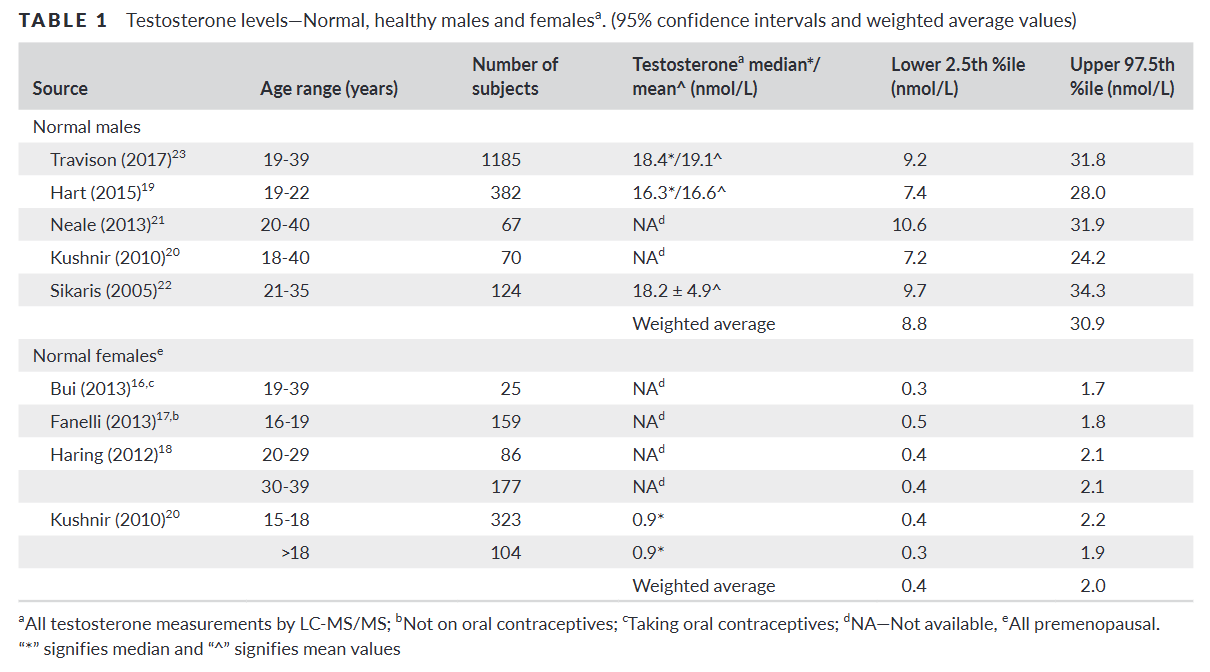
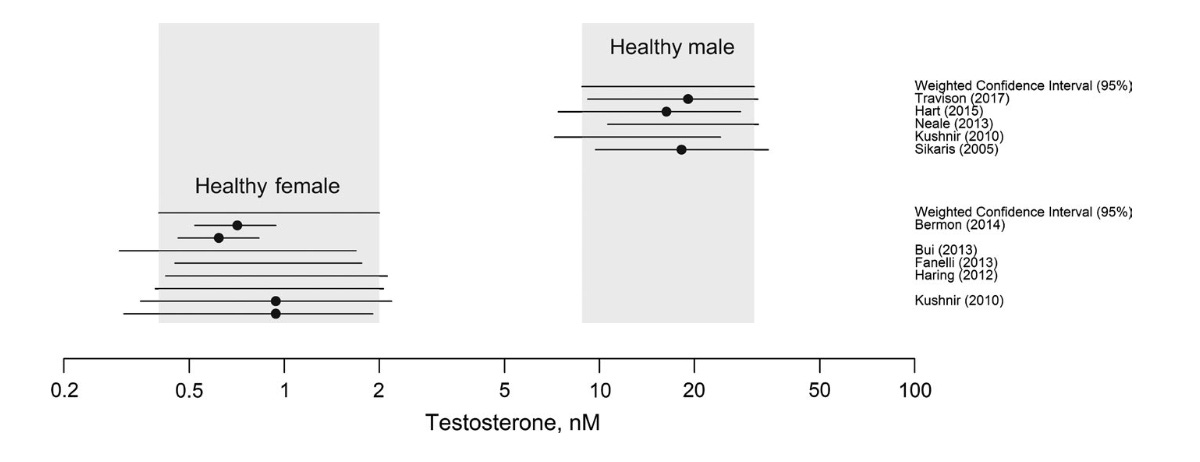
For instance, in 2018, Richard V. Clark of USADA (United States Anti-Doping Agency) and his colleagues published a review that summarized the “available data on testosterone levels in normal, healthy adult males and females”, with the goal of establishing a “physiologic reference framework”.4 The purpose of establishing such a framework, in the context of sex separation in elite sports, was to evaluate individuals who are intersex or have conditions that affect their testosterone levels.
Clark and his colleagues looked at data from the peer-reviewed literature, taking into account only the most accurate measurements. Indeed, there are different methods—called assays—for measuring testosterone. The most reliable assays measure testosterone in serum, and the gold standard among these is LC-MS/MS, which is more reliable than the immunoassays routinely used in medical blood work.5 This difference is especially important for the measurement of testosterone in women, as they have very low concentrations of the hormone compared with men.
Clark and his colleagues found “a clear bimodal distribution” of total testosterone levels in males and females.6 The weighted average of several studies produced the following ranges, spanning from the lower 2.5 to the upper 97.5 percentile:
Total testosterone in males: 8.8 to 30.9 nmol/L.
Total testosterone in females: 0.4 to 2.0 nmol/L.
This means that, in the healthy state, the lower end of the male range (that includes 95% of all males) is four to five times higher than the upper end of the female range (that includes 95% of all females). There is indeed a clear gulf separating the two distributions of total testosterone.
💥 Hypogonadism and hyperandrogenism
Men whose testosterone levels are too low—i.e., outside the “normal” male range—tend to experience negative symptoms, including fatigue, low energy, lack of motivation, brain fog, low libido, absence of nocturnal erections, and poor body composition. This condition is known as hypogonadism, and it’s associated with a number of diseases.7
Due to differences in sensitivity to androgens between individuals, there is no fixed threshold to define hypogonadism in men.8 Moreover, different symptoms can kick in at different levels.9 Nevertheless, various international medical societies, such as the Endocrine Society and the American Urological Association, rely in their guidelines on a threshold value. This value is usually set at the lower end of the “normal” male range in healthy adults, and it varies across guidelines from 9.2 to 12.1 nmol/L.10
As you might have noticed, these threshold values are much higher than the upper end of the “normal” female range of testosterone (~ 2.0 nmol/L). Furthermore, research that looks at symptomatic men finds that hypogonadism occurs at even higher thresholds than the ones set in the guidelines.11
Something analogous to men’s hypogonadism happens in women whose androgen levels—notably testosterone—are consistently too high—i.e., outside the “normal” female range. These women experience negative symptoms that include excess facial or body hair (hirsutism), acne, hair loss, and menstrual irregularities. Other symptoms, such as deepening of the voice, clitoral enlargement (clitoromegaly), and reduction of breast size, can happen in extreme cases. The condition of excess androgens in women is called hyperandrogenism, and it’s also associated with a number of diseases.12
The levels at which these negative symptoms appear in women are far, far lower than the lower end of the “normal” male range.13 Hyperandrogenism mostly happens in women who have polycystic ovarian syndrome (PCOS), the majority of whom show elevated testosterone levels compared with healthy females.14 In the overwhelming majority of women with PCOS, testosterone levels don’t exceed 4 nmol/L, which is much lower than the lower end of the “normal” male range of testosterone (~ 8.8 nmol/L).15
To summarize, not only is there a large gap in testosterone distributions between men and women, but slightly going out of one distribution in the direction of the other spells medical trouble for each respective sex, well before getting anywhere close to the other distribution.16
❓ Origin
In the face of such clear-cut evidence refuting the claim of a “considerable overlap” in testosterone levels between the sexes, how could such a ridiculous claim have originated?
A possible origin for this idea is research in the social sciences that measures testosterone in saliva. In the original paper by the five professors of psychology, most of the articles cited in support of the claim relied on measuring testosterone in saliva rather than in blood, even though these articles were not concerned with comparing testosterone levels between men and women.
Data from salivary testosterone assays do show some overlap in the distributions between men and women,17 which might be how Sari van Anders came to believe in the “considerable overlap”. This fact alone, however, is a testament to how grossly inaccurate salivary testosterone assays are as a measurement method. Indeed, salivary assays correlate poorly with serum assays, especially when the concentration of testosterone is very low, as is the case in women.18 A salivary assay can show that some women’s testosterone levels are apparently higher than some men’s, whereas in reality—when they are accurately measured in serum with LC-MS/MS—the opposite is true.
Sari van Anders herself frequently uses salivary testosterone to investigate research questions of how social factors might influence the levels of this hormone in humans. Some of her published work purports to have found that baby cries decrease testosterone in men, that merely “performing gender” by acting in stereotypical ways increases testosterone in women, and that being single or polyamorous is associated with higher testosterone in both men and women.19 But due to the inaccuracy of salivary testosterone, these research findings are, in my humble opinion, basically useless.
📌 Conclusions
Academics who continue to propagate the bizarre claim of a “considerable overlap” in testosterone levels between men and women ought to familiarize themselves with the literature on testosterone assay validation, lest they too immortalize their embarrassment in the academic record.
Furthermore, if social scientists are serious about studying the way social factors and “gender” might influence testosterone levels in humans, they should spend the extra money from their research funds and measure this hormone in serum with the gold standard LC-MS/MS, instead of relying on grossly inaccurate data from mouth swabs. Rigorous measurement may prove perilous, however, as it risks invalidating that entire field of study.
🎁 Bonus
The 2018 paper also made the claim that “average levels of estradiol […] do not differ between women and men”. This claim is also demonstrably false.20 Estradiol levels in women vary throughout the menstrual cycle, reaching their lowest during the early follicular phase and their highest before ovulation. In contrast, estradiol levels in men are relatively stable. Taking this into account, the median estradiol concentration of premenopausal women is anywhere from 1.8 to 3.6 times that of men.21
👍 Leave a like on this post and tell me your thoughts about it in the comments.
Hyde, Janet S., et al. (2018). The Future of Sex and Gender in Psychology: Five Challenges to the Gender Binary. American Psychologist, 74(2), 171–193.
For instance, the claim, along with several other bizarre ones, is repeated in p. 5 of van Anders, Sari M. (2024). Gender/Sex/ual Diversity and Biobehavioral Research. Psychology of Sexual Orientation and Gender Diversity, 11(3), 471–487.
Hyde et al., op. cit., p. 174.
Clark, Richard V., et al. (2019). Large divergence in testosterone concentrations between men and women: frame of reference for elite athletes in sex‐specific competition in sports, a narrative review. Clinical Endocrinology, 90(1), 15–22.
Matsumoto, Alvin M., & Bremner, William J. (2004). Editorial: Serum Testosterone Assays—Accuracy Matters. The Journal of Clinical Endocrinology & Metabolism, 89(2), 520–524.
Total testosterone refers to all testosterone in blood plasma, most of which circulates in bound form. It is bound to proteins that facilitate transportation. Testosterone is mostly bound to sex hormone-binding globulin (SHBG), but some of it is bound to albumin. Testosterone dissociates from these carrier proteins at the tissue level to be able to bind to androgen receptors and thus have its effects.
Overall, only a very small portion of total testosterone—less than 2%—is unbound in blood plasma. This portion is called free testosterone.
Another name—bioavailable testosterone—refers to free testosterone plus the small portion of the hormone that is bound to albumin, because this binding is considered weak.
Reference: Nieschlag, Eberhard, Behre, Hermann M., & Nieschlag, Susan (Eds.). Testosterone: Action, Deficiency, Substitution (4th ed.). Cambridge University Press.
Basaria, Shehzad. (2014). Male hypogonadism. The Lancet, 383(9924), 1250–1263.
In particular, the number of CAG repeats in the androgen receptor gene has been implicated in the issue of sensitivity. See, for example, Zitzmann, Michael, & Nieschlag, Eberhard. (2003). The CAG repeat polymorphism within the androgen receptor gene and maleness. International Journal of Andrology, 26(2), 76–83.
Zitzmann, Michael, et al. (2006). Association of Specific Symptoms and Metabolic Risks with Serum Testosterone in Older Men. The Journal of Clinical Endocrinology & Metabolism, 91(11), 4335–4343.
Salter, Carolyn A., & Mulhall, John P. (2019). Guideline of guidelines: testosterone therapy for testosterone deficiency. BJU International, 124(5), 722–729.
For example, a threshold of 13.9 nmol/L was found to be more appropriate for defining hypogonadism in Scovell, Jason M., et al. (2015). Hypogonadal symptoms in young men are associated with a serum total testosterone threshold of 400 ng/dL. BJU International, 116(1), 142–146.
Azziz, Ricardo et al. (2009). The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertility and Sterility, 91(2), 456–488.
For instance, a cutoff value of 1.57 nmol/L is chosen to define hyperandrogenism in Pasquali, Renato, et al. (2016). POSITION STATEMENT: Defining Hyperandrogenism in Women With Polycystic Ovary Syndrome: A Challenging Perspective. The Journal of Clinical Endocrinology & Metabolism, 101(5), 2013–2022.
Azziz, Ricardo, et al. (2006). Criteria for Defining Polycystic Ovary Syndrome as a Predominantly Hyperandrogenic Syndrome: An Androgen Excess Society Guideline. The Journal of Clinical Endocrinology & Metabolism, 91(11), 4237–4245.
See, for example, Keefe, Candance C., et al. (2014). Simultaneous Measurement of Thirteen Steroid Hormones in Women with Polycystic Ovary Syndrome and Control Women Using Liquid Chromatography-Tandem Mass Spectrometry. PLOS One, 9(4), e93805.
The difference is even greater for free testosterone—the small portion that is not bound to carrier proteins. Indeed, the proportion of total testosterone that is free in women’s blood is smaller than in men’s, because women have, on average, higher levels of sex hormone-binding golublin (SHBG).
See, for example, Handelsman, David J., et al. (2016). Estimating age-specific trends in circulating testosterone and sex hormone-binding globulin in males and females across the lifespan. Annals of Clinical Biochemistry, 53(3), 377–384.
See, for example, Granger, Douglas A., et al. (2004). The “trouble” with salivary testosterone. Psychoneuroendocrinology, 29(10), 1229–1240.
For example, in a sample of 20 women in Granger et al. (2004), the correlation between testosterone measured in saliva and free testosterone measured in serum was only r = 0.37, and it didn’t reach statistical significance.
All these works may be found on her Google Scholar profile.
A position statement from the Endocrine Society, for instance, reads: “Estradiol levels in men are lower than in premenopausal women.” Rosner, William et al. (2013). Challenges to the Measurement of Estradiol: An Endocrine Society Position Statement. The Journal of Clinical Endocrinology & Metabolism, 98(4), 1376–1387.
For rigorous measurement of estradiol in the adult population with LC-MS/MS, see, for example, Frederiksen, Hanne, et al. (2020). Sex-specific Estrogen Levels and Reference Intervals from Infancy to Late Adulthood Determined by LC-MS/MS. The Journal of Clinical Endocrinology & Metabolism, 105(3), 754–768.